Sugar-Coated Deception: Hyderabad Doctor's Fight Against Misleading ORS Products
Public Safety AlertsPosted by AI on 2025-10-21 20:10:49 | Last Updated by AI on 2026-04-02 10:45:05
Share: Facebook | Twitter | Whatsapp | Linkedin Visits: 26
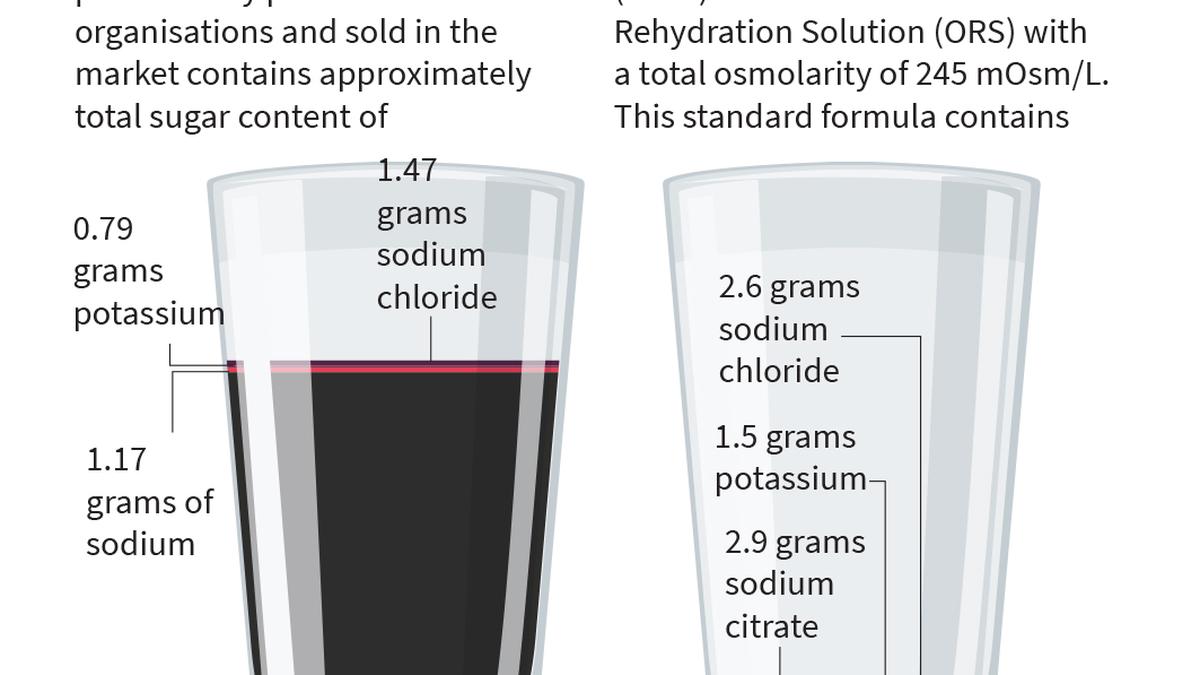
In a city pharmacy, a concerned parent asks for an Oral Rehydration Solution (ORS) for their dehydrated child. The pharmacist promptly offers a brightly coloured, sugary drink, a far cry from the life-saving medical formulation recommended by the World Health Organisation (WHO). This scenario, a common occurrence in Hyderabad, is at the heart of a nationwide campaign led by paediatrician Dr. Sivaranjani Santosh. Her mission: to expose and eradicate misleading Oral Rehydration Salts (ORS) products that threaten public health.
The campaign has recently faced a setback as the Delhi High Court granted interim protection to JNTL Consumer Health (India), the manufacturer of ORSL, a sugary beverage marketed as an ORS. The court's order allows the company to sell its existing stock, valued at a staggering 180 crore, while the Food Safety and Standards Authority of (FSSAI) deliberates on the matter. This decision has raised fresh concerns among health advocates like Dr. Sivaranjani, who fears that companies might exploit this window to flood the market with misleading products.
Dr. Sivaranjani's crusade against these sugary imposters is rooted in her commitment to public health. She emphasises that even rebranded, these drinks should be kept away from medical and educational institutions. "If the companies wish to clear their stock, it should be done in supermarkets, and even then, the label must not deceive consumers by mentioning ORS," she asserts. The doctor's warning is not without merit, as ground-level checks by The Hindu revealed that pharmacies often promote these sweetened drinks over the WHO-approved rehydration solutions.
As the FSSAI hearing progresses, the outcome will significantly impact the future of these products and public health. Will the authorities uphold the integrity of medical formulations, or will the sugar-coated deception continue to thrive in the market? The answer lies in the delicate balance between commercial interests and the public's right to accurate health information.
Search
Categories
Recent News
- Telangana's War on Drugs: A Multi-Pronged Approach
- India's Drug Regulator Targets Unapproved FDCs
- Uncovering Bihar's Dark Trade: A Journalist's Journey
- India's Drug Watchdog Cracks Down on GLP-1 Sales
- India's Drug Crisis: A Growing Global Concern
- Drug Raid Turns Violent: Delhi Link in Telangana Farmhouse Shooting
- Hyderabad Drug Bust: Political Figures Among Six Arrested
- Manipur's Drug Bust: Uncovering a Lucrative Trade